



The report offers an in depth analysis on Global Alpha Thalassemia Market 2022 that involves many as

Pre-open movers

The week in pharma: action, reaction and insight – week to September 23

This biotech has made tremendous progress recently.

The FDA approved a tumor agnostic treatment for cancer, a lower dose MRI contrast agent, the first generic of Tazorac gel, and a gene therapy for a rare disease. Advisory committees vote down poziotinib for NSCLC and Pepaxto for multiple myeloma and give positive vote for microbiotic-based C. diff t…

The U.S. Food and Drug Administration (FDA) has approved Bluebird bio’s gene therapy for the treatment of a rare neurological disorder, the company said late on Friday.
/cloudfront-us-east-2.images.arcpublishing.com/reuters/OV2PC52TJZNG5PW4JK4QPO2EU4.jpg)
“SKYSONA is the first FDA approved therapy shown to slow the progression of neurologic dysfunction in boys” with early, active Cerebral Adrenoleukodystrophy (CALD), the company said in a statement, saying CALD is a “devastating and fatal neurodegenerative disease.” U.S. seeing decline in monkeypox…

Upon taking the helm at precision oncology-focused Scorpion Therapeutics last summer, Axel Hoos told Endpoints News that he had a clear vision of the type of leaders he wanted to hire. It appears that former Sanofi project head Michael Streit, one of Hoos’ old colleagues from GSK, fits the bill.

The market is struggling, yet biotech stocks are acting like it’s good times again. Here’s why.

Overview: The recombinant adeno-associated virus (AAV) is an exciting new gene delivery vector. The development of this vector was facilitated by an

DUBLIN--(BUSINESS WIRE)--Sep 15, 2022--

Genes are composed of deoxyribonucleic acid, which contain essential information for producing proteins that are crucial for the optimal functioning of the body
The Institute for Clinical and Economic Review (ICER) concluded that even at a hefty lifetime cost of $2.5 million, gene therapies for hemophilia A and B are worth it.
<p>The Spanish biotech, formed in 2011, is preparing for the potential European launch of leriglitazone for X-linked adrenoleukodystrophy after the EMA accepted its file.</p> <br class=“t-last-br” />

The market is struggling, yet biotech stocks are acting like it’s good times again. Here’s why.

Premarket Mover: bluebird bio Inc (BLUE) Up 4.37%

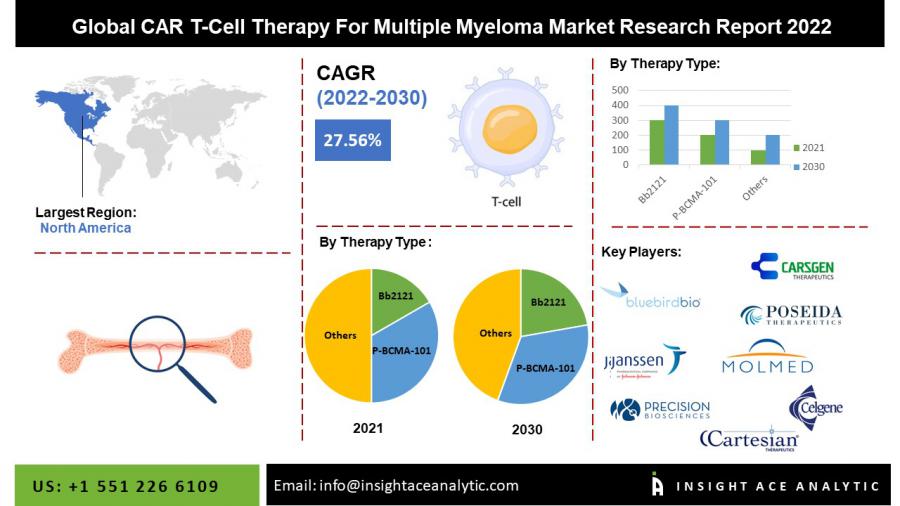
Global CAR T-Cell Therapy For Multiple Myeloma market is expected to show a CAGR of 27.56% during a forecast period of 2022-2030.
Coherent Market Insights has published a new research report titled “Personalized Cell Therapy Market 2022 analysis by Market Trends (Drivers, Constraints,

“Cell and Gene Therapy” Market Insights | No of Pages111| By Appliication , By Type . Cell and Gene Therapy Market Report offers a
Think twice before investing in these companies.
